NYU scientists use ScanImage to monitor dopamine-induced brain activity in a model of Parkinson’s disease
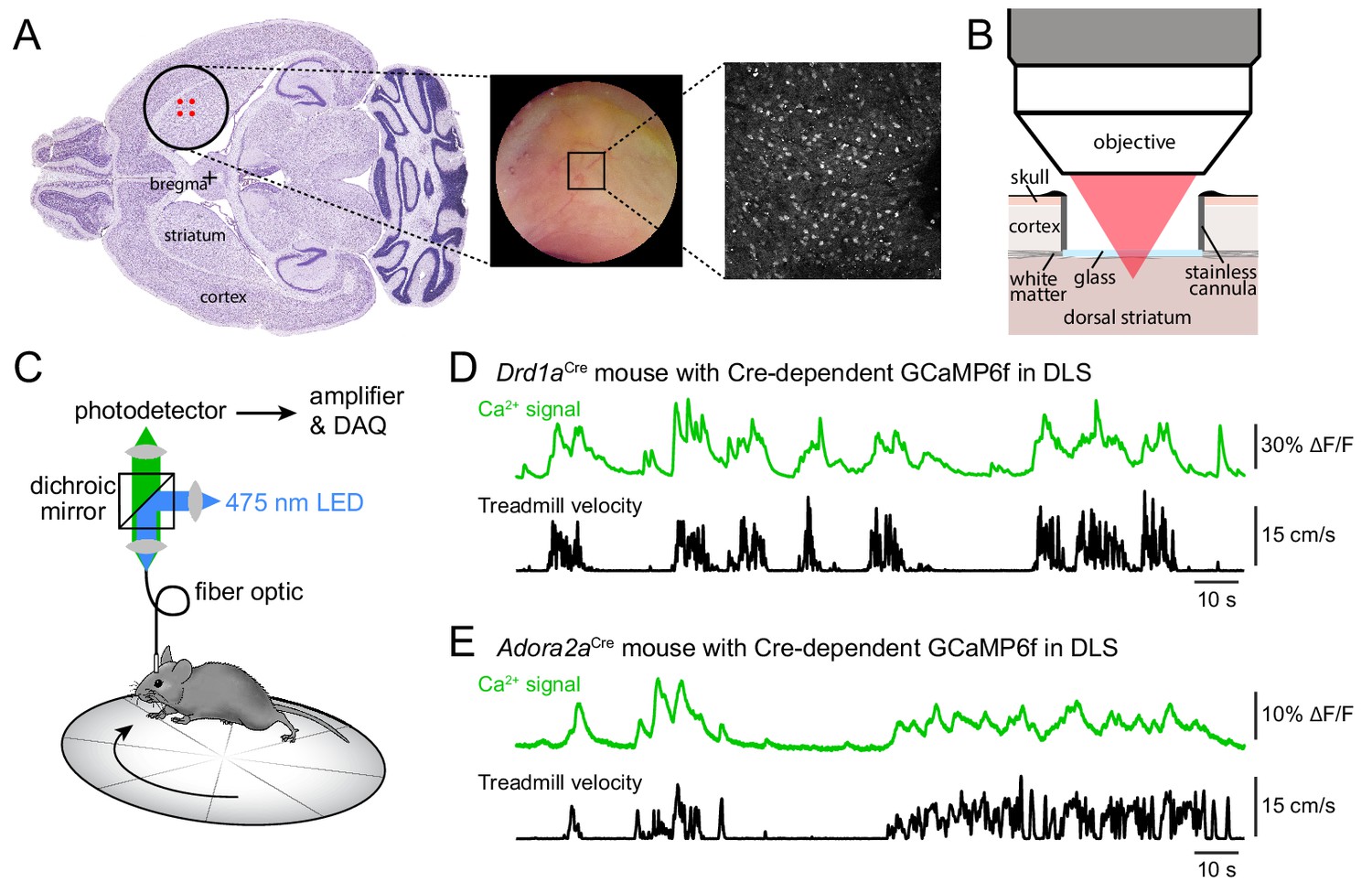
Humans and animal species can move in controlled motion sequences because of a delicate balance in the signaling of certain neurons in the area of the brain called the striatum. In this brain region, some neurons tell muscles to move, while others tell muscles to hold steady. In the brains of patients with Parkinson’s disease, more of the neurons that signal movement are activated resulting in a disruption of the delicate balance, which in turn disrupts voluntary movement. Using ScanImage software, researchers at New York University found evidence supporting dopamine’s integral role in this process.
Dopamine (DA) is well known to enable nerve cells to communicate with each other in the brain. In an effort to go deeper into understanding this process, the research team led by Dr. Marta Maltese, used two-photon calcium imaging to monitor activity in the brains of mice. As the mice ran on a treadmill, the scientists observed activity among the two types of neurons — the ones that incited movement, and the ones that directed a muscle to be still.
“This research is amazing because the activity of the neurons that create a balance between moving a muscle and not moving a muscle can be seen through the microscope while the mouse is moving on the treadmill,” says MBF Bioscience Research Liaison Dr. Dan Peruzzi. “Not only could they see the activity of the neurons, they could also differentiate which neurons activate the ‘do not move the muscle’ instructions and which neurons send signals to ‘move the muscle’.
The researchers found that when the mouse was on the treadmill, an equal number of both types of neurons were activated. But when they introduced drugs that reduced dopamine levels, mimicking what happens in Parkinson’s disease, the balance was upset — more of the neurons that tell a muscle not to move were activated.
“Our imaging approach therefore offers the ability to simultaneously monitor and compare the activity of hundreds of striatal neurons (mean ± SEM: 327 ± 13 per field of view, range: 131–442) belonging to both direct and indirect pathways with high spatial resolution during a simple behavior… Our results reveal that, in addition to its effects on firing rates, DA regulates striatal output by reconfiguring its movement-related ensemble code.” (Maltese et.al., 2021)
Maltese, M., March, J. R., Bashaw, A. G., & Tritsch, N. X. (2021). Dopamine differentially modulates the size of projection neuron ensembles in the intact and dopamine-depleted striatum. eLife, 10, e68041. https://doi.org/10.7554/eLife.68041