
Delayed loss of neurons occurs in mice with mild TBI and anxiety
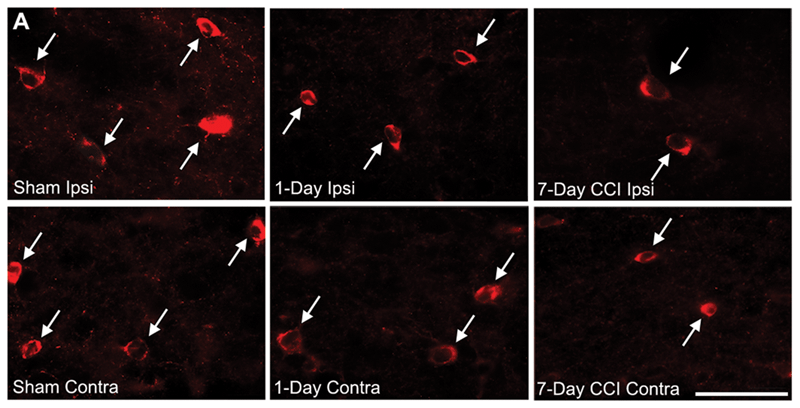
Almeida-Suhett et al saw delayed loss of GABAergic interneurons in the BLA within the first week after mild CCI. (Representative photomicrographs of GAD-67 immunohistochemically stained GABAergic interneurons in the BLA of sham (left), 1-day CCI (middle), and 7-day CCI (right) animals. Total magnification is 630x; scale bar, 50 µm.)
Soldiers, athletes, and other individuals who suffer a traumatic brain injury often develop anxiety disorders, but scientists aren’t sure why. Some speculate that fear about future health or the stress of the trauma itself contributes to elevated anxiety, while others suspect changes happening inside the brain as a result of the injury are to blame.
Researchers at Maria Braga’s lab at the Uniformed Services University of the Health Sciences in Bethesda, Maryland, recently found direct evidence that physical changes happen in the brain after TBI that coincide with increased anxiety levels.
She and her team studied a rat model of mild TBI, focusing on the basolateral amygdala (BLA) – a brain region often damaged by TBI, which has also been associated with increased fear and anxiety in instances of hyperactivity.
To find out what happens in the BLA that might be causing anxiety after a mild TBI, the researchers analyzed changes in synaptic activity in this region. Using Stereo Investigator with the optical fractionator probe to perform a stereological quantification of Nissl-stained and GAD-67 immunostained brain cells, they found that many of the inhibitory neurons – the cells that quiet activity – were lost seven days after injury. Whole cell recordings from principal neurons confirmed that the inhibitory cells’ synaptic transmissions were impaired during this period, resulting in increased excitability and “open field tests” showed elevated anxiety levels in post-injury rats at the exact same time point.
“These results suggest that mTBI causes not only a significant reduction in inhibition in the BLA, but also an increase in neuronal excitability, which may contribute to hyperexcitability and the development of anxiety disorders,” the authors say in their paper, adding that their results “clearly demonstrate that the increase in anxiety-like behaviors seven and 30-days after mTBI was due to the [controlled cortical impact] and not due to uncontrolled stress or the surgery.”
Comments about the unbiased stereology methods in this paper by Dr. Dan Peruzzi, Staff Scientist
Reporting the parameters used in a stereological study is very important for reproducibility. The authors did an exemplary job at this for the optical fractionator probe employed by them to estimate the number of neurons and the number of GAD-positive neurons in the basal lateral amygdala. Guidelines for reporting parameters can be found on stereology.info.
The portion of the methods section that describes the parameters should start with a description of how the region is defined. In this case the Atlas used is given. Differences in anatomical borders are often at the root of discrepancies among the results of different studies.
The fraction of tissue that is sampled should be given next. We are told the section sub-fraction (one out of five), the area sub-fraction (the counting frame area, 35 X 35 microns, divided by the grid-step, 190 X 190 microns), and the height sub-fraction (the disector height, 12 microns for neurons and 20 microns for GAD-positive cells, divided by the average mounted section thickness, 24 microns – measured every five sections). The reason a smaller disector height was used for neurons is likely that the Nissl stain resulted in more variation in tissue thickness after histology than the immunohistochemistry caused in the sections labeled for GAD. In other words, the Nissl stained sections for neurons had a smaller minimum thickness than the immonohistochemically labeled sections for GAD-positive neurons; and the minimum thickness of the sections will restrict the height of the disector. Reporting the range of tissue thickness would help explain the need for different height disectors.
The optical fractionator probe can’t be executed properly without many focal planes, so it is good that the sections have a mean of 24 microns thickness, but an objective with good resolving power must also be used. That means using an objective that is 40x, 60x, 63x or 100x and has a 1.3 or 1.4 numerical aperture; a rule for the optical disector that is frequently broken. Many papers either report using the incorrect objective, or don’t state what objective was used at all. In this case we were very happy to see the authors describe using a 63x oil immersion objective.
All of the other parameters needed to understand and reproduce the study are given in the methods section. The guard zone height is two microns, however no explanation of why is given. The leading edge that is being used to count the particles is identified: in the case of Nissl stained neurons it is the leading edge of the soma and in the case of immuno-labeled GAD-positive cells, it is the ‘top’ of the nucleus that is used to decide if the particle is in the disector or not. The average number of leading edges (particles) counted per section is given, but the range is not. Regarding precision; it is explained that coefficients of error (CEs) are used to decide if enough sampling is done, and the CEs are identified (Gunderson, m = 1 and Schmitz-Hof) and their values given. The thorough and conscientious job done reporting parameters of this unbiased stereological study makes it possible for other researchers to trust and replicate the experiments.
Almeida-Suhett, C. P., Prager, E. M., Pidoplichko, V., Figueiredo, T. H., Marini, A. M., Li, Z., Eiden, L., Braga, M. F. M. (2014). Reduced GABAergic Inhibition in the Basolateral Amygdala and the Development of Anxiety-Like Behaviors after Mild Traumatic Brain Injury. Plos one, 9(7), e102627. doi: 10.1371/journal.pone.0102627
.


