Scientists Discover Anorexia-Driven Changes to Dendrites With Neurolucida
Gaunt facial features and a frighteningly thin figure are physical hallmarks of anorexia nervosa, an eating disorder that predominantly affects adolescent girls. But in addition to extreme weight loss, changes take place that aren’t as visually apparent. For the first time, scientists in New York have found evidence of brain plasticity in the activity-based anorexia (ABA) mouse model.
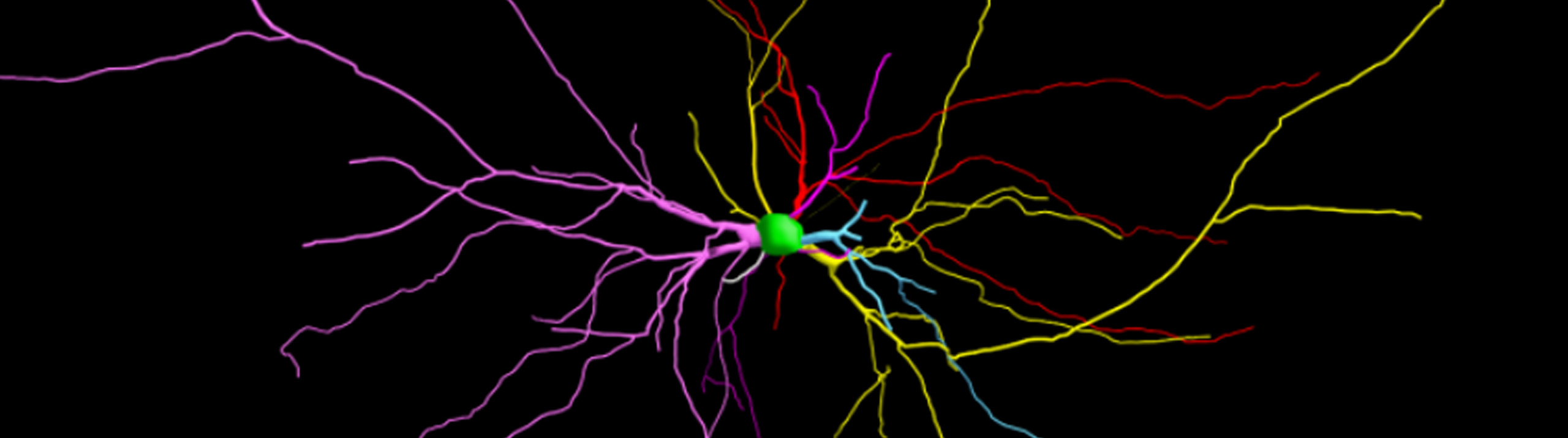
Led by Dr. Chiye Aoki of New York University, the research team used Neurolucida to analyze pyramidal neurons in the rat brain. Since anorexia is linked to elevated stress hormones and anxiety, the researchers focused on the hippocampus, a region that regulates anxiety and is known to change structurally in response to hormones and stress.
“Using Neurolucida, we were able to collect, store, and analyze large amounts of data with more precision and accuracy than would have been possible without the digital interface,” said Tara Chowdhury, a graduate student working in Dr. Aoki’s lab, and first author of the paper.
“Additionally, with its very approachable interface, the software allowed us to trace dendrites, get precise thickness measurements, and categorize spine types easily during tracing. The built-in Sholl analysis and spine analysis tools resulted in quick quantification of all the measurements that would have taken hours to achieve without Neurolucida.”
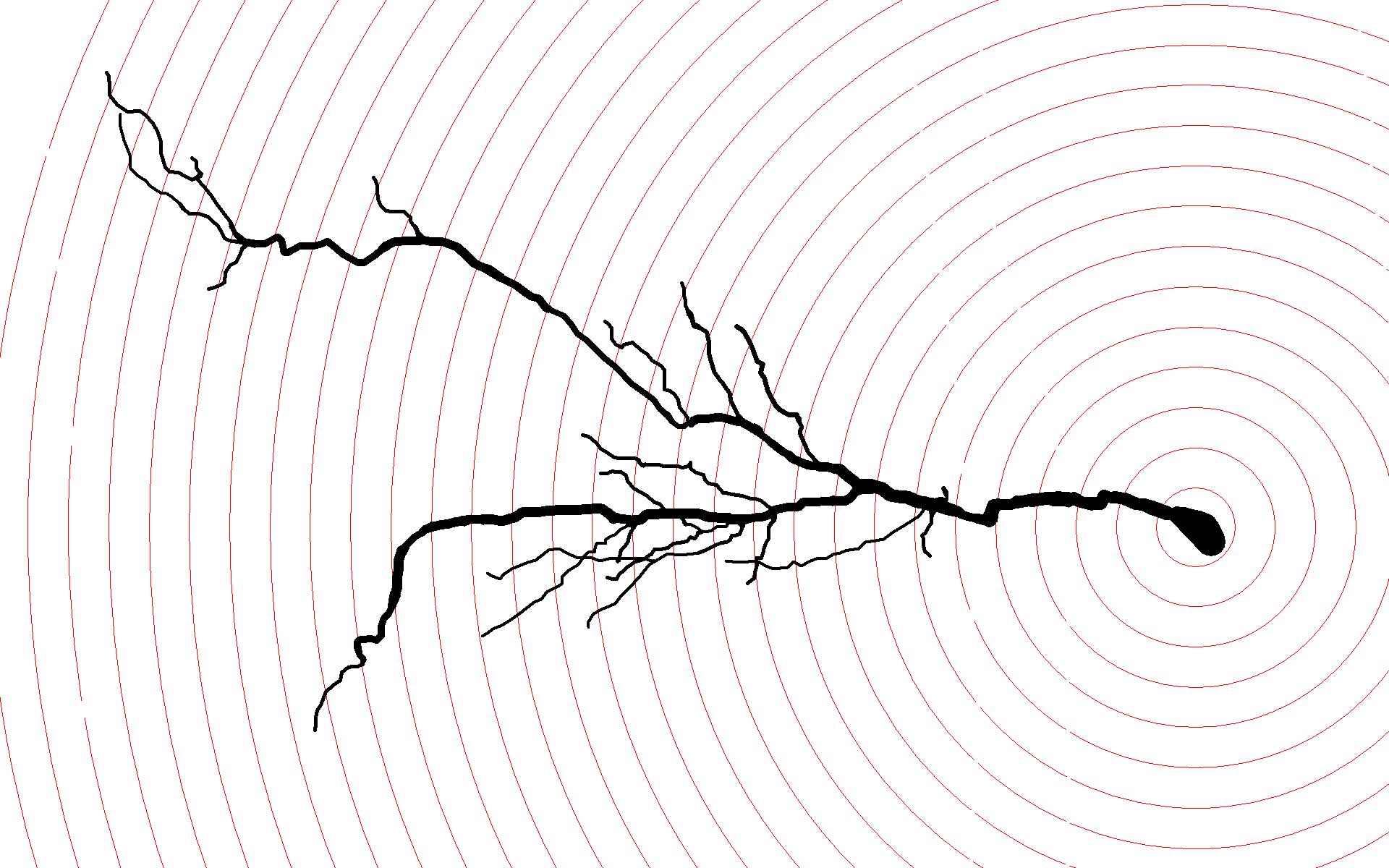
A digital reconstruction of a CA1 pyramidal cell from the ventral hippocampus of a rat with activity-based anorexia, traced using Neurolucida with Sholl spheres at 20 micron intervals. Cells in this region featured greater dendritic length and branching versus controls.
Using the Sholl analysis technique with Neurolucida to analyze Golgi-stained neurons, the researchers observed structural differences in various parts of the ABA rat hippocampus. These rats displayed neurons with shorter dendrites and less branching in an area called the stratum radiatum (SR) in the dorsal hippocampus, compared to controls, but neurons in the SR of the ventral hippocampus featured greater dendritic length and branching versus controls. In other words, one area displayed neural atrophy, while another area showed growth – changes observed after a remarkably short period of four days.
By comparing four experimental groups – control (ad libitum food access, no wheel access), ABA (1 h per day food access, ad libitum wheel access), exercise control (ad libitum food and wheel access), and food restriction control (1 h per day food access, no wheel access) – the scientists had enough evidence to deduce that food restriction and exercise separately influence different regions of the hippocampus.
“Wheel running is driving the [decreased branching] in rostral-dorsal SR,” they say, while “food restriction increased dendritic branching of the ventral region.” Though the paper also points out that food restriction decreased dendritic length in both the dorsal and ventral region in an area called the stratum lacunosum-moleculare (SLM).
The scientists speculate that the physical changes may be a result of increased or decreased excitability of the neurons in these areas, with increased excitability causing dendritic growth, and vice versa.
“Synaptic inputs via the perforant path and Schaffer collaterals may be playing independent roles in remodeling pyramidal cell apical arbors, shaping their firing structure, and, ultimately, influencing behavior,” they say in their paper, adding that “these results indicate that ABA elicits pathway-specific changes in the hippocampus that may underlie the increased anxiety and reduced behavioral flexibility observed in ABA.”
Chowdhury, T., Barbarich-Marsteller, N., Chan, T., & Aoki, C. (2013). Activity-based anorexia has differential effects on apical dendritic branching in dorsal and ventral hippocampal CA1. Brain Structure and Function, 1-11. doi: 10.1007/s00429-013-0612-9.


